I. Introduction
If you’ve ever taken a chemistry class, you’ve encountered the concept of molar mass. For nitrogen, that number—28.014 g/mol—might seem like just another fact to memorize.
But in the real world, nitrogen’s molar mass matters. It’s the foundation for calculations that engineers, technicians, and plant operators use every day. It determines how much nitrogen fits in a tank, how fast it flows through a pipe, and how much energy is needed to compress it.
This guide explains what nitrogen molar mass is, why it’s important, and how to use it in practical applications—from sizing storage tanks to calculating gas consumption.
II. What Is Nitrogen Molar Mass?
Let’s start with the basics.
Definition:
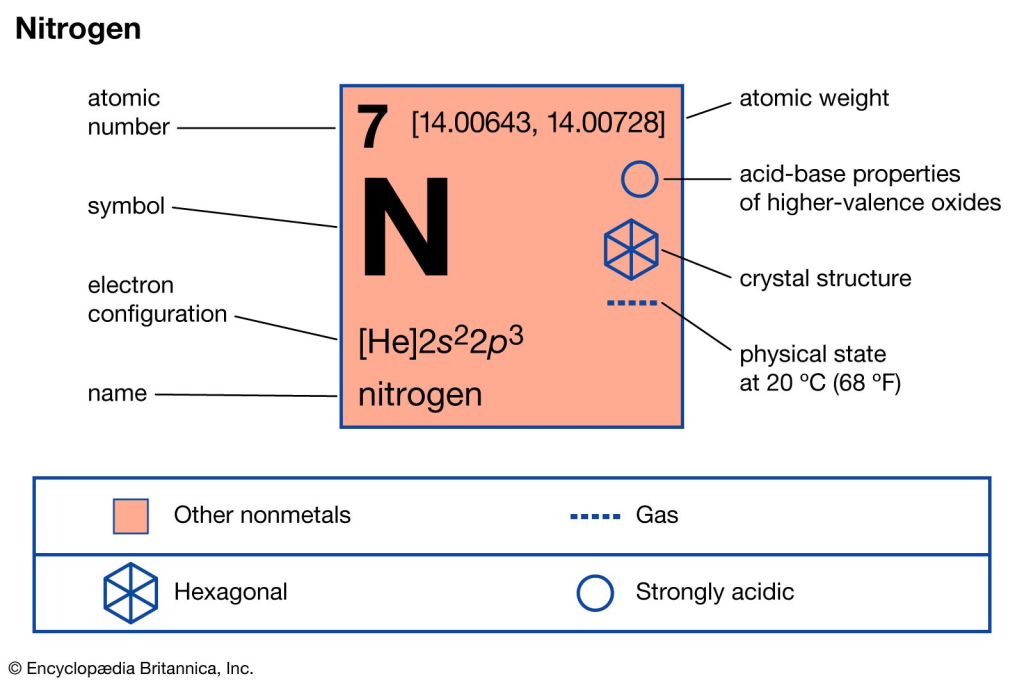
Molar mass is the mass of one mole of a substance. A mole is Avogadro’s number of particles—approximately 6.022 × 10²³ molecules.
For nitrogen (N₂):
- Atomic mass of nitrogen (N): 14.007 g/mol
- Nitrogen gas is diatomic (N₂): 2 × 14.007 = 28.014 g/mol
The numbers you’ll see:
- 14.007 g/mol: Mass of a single nitrogen atom
- 28.014 g/mol: Mass of a nitrogen molecule (N₂ gas)
- 28.0 g/mol: Common rounded value for practical calculations
Why it’s not exactly 28:
Natural nitrogen is a mix of isotopes. Nitrogen-14 is most common (99.6%), with trace amounts of nitrogen-15. The weighted average gives 14.007 per atom, 28.014 per molecule.
III. Why Nitrogen Molar Mass Matters in Real-World Applications
This isn’t just a chemistry textbook number. Molar mass affects real engineering decisions.
Gas density:
The density of nitrogen gas depends on its molar mass. At standard temperature and pressure (0°C, 1 atm), nitrogen density is about 1.25 g/L.
Why it matters:
- Storage sizing: Knowing density tells you how much nitrogen fits in a tank
- Pipeline design: Density affects pressure drop calculations
- Buoyancy: Nitrogen is slightly lighter than air (28.014 vs. 28.96 g/mol for air)
Flow calculations:
Mass flow rate, volumetric flow rate, and pressure are connected through the ideal gas law—which uses molar mass.
Compression energy:
The energy required to compress nitrogen depends on its specific gas constant, which is derived from molar mass.
Gas mixtures:
When blending gases (e.g., for food packaging or medical applications), molar mass helps calculate proportions.
IV. How to Calculate Nitrogen Molar Mass
For those who need the math.
From atomic weights:
Nitrogen molar mass (N₂) = Atomic mass N × 2
= 14.0067 g/mol × 2
= 28.0134 g/mol
Standard accepted value:
For most calculations, 28.0134 g/mol or simply 28.0 g/mol is used.
Isotopic composition:
- ¹⁴N: 99.636% abundance, mass 14.003074 u
- ¹⁵N: 0.364% abundance, mass 15.000108 u
Weighted average: (0.99636 × 14.003074) + (0.00364 × 15.000108) = 14.0067 u per atom
For practical engineering:
Use 28.0 g/mol for most calculations. The 0.05% error is negligible for equipment sizing. For precise scientific work, use 28.0134 g/mol.
V. Using Molar Mass in Gas Calculations
Here’s where the number becomes useful.
Ideal gas law:
PV = nRT, where n = mass / molar mass
Finding mass from volume:
Mass (kg) = (Pressure in Pa × Volume in m³ × Molar mass) / (Gas constant × Temperature)
Simplified for standard conditions (0°C, 1 atm):
1 mole of nitrogen = 22.414 L = 28.014 g
Quick reference:
| Quantity | Value |
| 1 mole N₂ | 28.014 g |
| 1 mole N₂ at STP | 22.414 L |
| 1 kg N₂ at STP | 0.800 m³ |
| 1 m³ N₂ at STP | 1.250 kg |
For compressed gas:
At 100 bar (10 MPa) and 20°C, nitrogen density is about 115 kg/m³—about 90 times denser than at atmospheric pressure. This is why high-pressure storage is efficient.
VI. Nitrogen Molar Mass vs. Air Molar Mass
Comparing nitrogen to air helps understand its behavior.
| Property | Nitrogen (N₂) | Air |
| Molar mass | 28.014 g/mol | 28.96 g/mol |
| Density at STP | 1.25 kg/m³ | 1.29 kg/m³ |
| Specific gas constant | 296.8 J/(kg·K) | 287.1 J/(kg·K) |
Implications:
- Buoyancy: Nitrogen is slightly lighter than air. In still conditions, nitrogen will rise slowly.
- Leak detection: Nitrogen leaks are harder to detect than heavier gases because they disperse quickly.
- Compression: The lower specific gas constant means slightly different compression characteristics.
For engineering:
Many calculations use air properties for rough estimates. For precise work, use nitrogen-specific values.
VII. Practical Applications of Nitrogen Molar Mass
Where this number actually matters.
Sizing storage tanks:
Need to store 500 kg of nitrogen at 200 bar? Using molar mass, you can calculate required tank volume.
Example:
- 500 kg N₂ = 500,000 g
- 500,000 g ÷ 28.014 g/mol = 17,850 moles
- At 200 bar and 20°C, volume = (nRT)/P
- Approximate: 0.5-0.6 m³ tank volume
Calculating purge gas requirements:
How much nitrogen to purge a 10 m³ tank? Multiply volume by density at operating pressure.
Estimating consumption:
If your process uses 100 m³/hr at 7 bar, what’s the mass flow? Use density at pressure to convert.
Cost calculations:
Knowing mass flow lets you calculate energy cost per kilogram of nitrogen produced or delivered.
FAQ
Q1: Why is nitrogen molar mass 28 and not 14?
A1: Nitrogen gas exists as diatomic molecules (N₂), not single atoms. Each molecule contains two nitrogen atoms, so the molar mass is twice the atomic mass: 2 × 14.007 = 28.014 g/mol.
Q2: Is nitrogen heavier or lighter than air?
A2: Nitrogen is slightly lighter than air. Air (mostly oxygen and nitrogen) has an average molar mass of about 28.96 g/mol, while nitrogen is 28.014 g/mol. The difference is small but measurable.
Q3: What’s the difference between nitrogen molar mass and molecular weight?
A3: They’re essentially the same. Molar mass (g/mol) is the modern term. Molecular weight (dimensionless) is the older term. Numerically, they’re identical.
Q4: How do I convert nitrogen volume to mass?
A4: Multiply volume (m³ at STP) by density (1.25 kg/m³). For example, 10 m³ at STP = 12.5 kg. For compressed gas, use density at your operating pressure and temperature.
Q5: Does nitrogen molar mass change with pressure?
A5: No. Molar mass is an intrinsic property of the substance. It doesn’t change with pressure, temperature, or volume. What changes is density, not molar mass.
Q6: Why do some sources say 28.0 and others 28.0134?
A6: 28.0 is rounded for convenience. 28.0134 is the precise value based on isotopic composition. For most engineering calculations, 28.0 is accurate enough. For scientific work, use the precise value.
Q7: How is nitrogen molar mass used in PSA generator design?
A7: PSA (Pressure Swing Adsorption) systems separate nitrogen from air. The molar mass difference between nitrogen and oxygen is small, but the kinetic properties—related to molecular size and mass—affect how quickly gases adsorb onto zeolite. Designers use these properties to optimize cycle timing.
Conclusion
Nitrogen molar mass—28.014 g/mol—might seem like a simple number from a chemistry textbook. But in the real world, it’s a fundamental tool for engineers, technicians, and plant operators.
It tells you how much nitrogen fits in a tank, how fast it flows through a pipe, and how much energy it takes to compress. It’s the bridge between the gas you measure in cubic feet and the mass you need to account for.
Whether you’re sizing storage, calculating consumption, or designing a system, understanding nitrogen molar mass helps you get the numbers right.
At MINNUO, we work with these numbers every day. From helping customers size generators to calculating gas requirements for specific applications, we rely on fundamental properties like molar mass to design systems that work. Because we know that accurate calculations lead to reliable results.


 sales2:+86 17506119168
sales2:+86 17506119168

